Pharmaceutical Giants Rush to Develop PD-(L)1 Bispecific Antibodies: A New Battlefield in Immunotherapy
In recent years, immune checkpoint inhibitors (ICIs) have emerged as a significant breakthrough in cancer therapy, reshaping traditional treatment paradigms. PD-1/PD-L1 pathway inhibitors have been widely used across various cancer treatments, demonstrating impressive efficacy. As PD-(L)1 inhibitor therapies continue to mature, pharmaceutical giants have turned their attention to PD-(L)1 bispecific antibodies (BsAbs), a new class of antibody drugs that has become a hot field for development in the industry.
The key advantage of PD-(L)1 bispecific antibodies is their ability to target both PD-1 and PD-L1 simultaneously, not only enhancing anti-tumor activity through stronger immune activation effects but also overcoming the limitations of single-target antibodies. As a result, pharmaceutical companies have invested heavily in developing PD-(L)1 bispecific antibodies, striving to achieve breakthroughs in this area.
PD-(L)1 bispecific antibodies are one of the brightest stars in antibody drug development. These bispecific antibodies can recognize two different antigens or targets at the same time, resulting in a synergistic effect. In their design, bispecific antibodies not only block the binding between PD-1 and PD-L1 but also recruit immune cells, enhancing the immune system's ability to attack tumors. This "two-pronged" strategy has made PD-(L)1 bispecific antibodies a focal point in cancer immunotherapy.
Currently, numerous pharmaceutical and biotechnology companies are actively advancing the clinical research of PD-(L)1 bispecific antibodies, especially in cancer immunotherapy, where they show significant promise. Some PD-(L)1 bispecific antibodies can not only target immune evasion mechanisms within the tumor microenvironment but also significantly improve patient survival, positioning them as the "new favorite" in cancer immunotherapy.
The PD-1/PD-L1 Pathway and Mechanism of Immune Escape
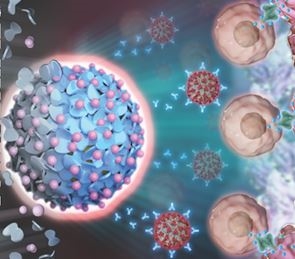
PD-1 (Programmed Cell Death Protein 1) is a crucial checkpoint in the immune system. By binding to its ligand PD-L1, PD-1 inhibits T-cell activation, regulating immune responses and preventing excessive immune reactions that could harm the body's tissues. However, tumor cells often exploit this mechanism to evade immune surveillance, promoting their growth and metastasis.
The role of the PD-1/PD-L1 pathway in immune evasion makes it a key target for immunotherapy. The application of PD-1 and PD-L1 monoclonal antibodies helps to relieve immune suppression, restore T-cell function, and boost the immune system's ability to recognize and eliminate tumor cells. As a result, immune checkpoint inhibitors are widely used in the treatment of various cancers, including non-small cell lung cancer, melanoma, and renal cell carcinoma.
Despite the promising clinical efficacy of PD-1/PD-L1 inhibitors, challenges remain. Some patients develop resistance to these therapies, and side effects, such as immune-related adverse events, can complicate clinical application. This has driven researchers and pharmaceutical companies to explore new treatment options, with bispecific antibodies emerging as a promising solution.
In the development of immunotherapy drugs, the use of cell models plays a critical role. Human PD-1 recombinant cell lines are among the most essential tools for studying the PD-1 pathway, widely used for drug screening, mechanistic research, and preclinical evaluation. By stably expressing the PD-1 protein in cells, researchers can simulate interactions between immune cells and tumor cells, explore the mechanisms of the PD-1/PD-L1 pathway, and evaluate the efficacy of PD-1/PD-L1 targeted therapies.
For example, using these recombinant cell lines, researchers can simulate immune escape processes in the tumor microenvironment, investigate the mechanisms of action of PD-1 inhibitors, and screen new antibody drugs. This tool is also crucial in evaluating the preclinical potential of drugs, contributing to the advancement of anti-tumor immunotherapies.
As the field of immunotherapy continues to evolve, the clinical application of PD-1/PD-L1 inhibitors has made significant strides. However, several challenges remain, particularly related to individual variation, resistance, and side effects. Researchers are actively exploring combination therapies to enhance treatment outcomes, such as combining PD-1 inhibitors with chemotherapy, targeted therapies, or vaccines. This may help overcome resistance and improve the overall efficacy of treatment.
Furthermore, as new immunotherapy strategies emerge, the application of PD-1 and related treatments may extend beyond cancer. Immune checkpoint inhibitors are showing promise in autoimmune diseases, infectious diseases, and other areas, making them a key focus in future medical research.
From the early days of single-target therapies to the current focus on bispecific antibodies, immunotherapy continues to innovate, transforming cancer treatment approaches. With the emergence of PD-1 recombinant cell lines and new immunotherapy solutions, we can look forward to a new era in cancer therapy, where more patients will benefit and the full potential of immunotherapy will be unlocked.